
Research and development expenses for the first quarter of 2022 were $5.1 million compared to $3.6 million for the same period in 2021. Net loss and net loss per share for the quarter ended Mawere $8.5 million and $1.18, respectively, compared to $5.4 million and $10.55, respectively, for the first quarter of 2021. Net cash used in operations for the quarter ended Mawas $7.1 million compared to $6.3 million for the same period in 2021. Cash balance does not reflect $15 million raised in April from PIPE financing. The decrease was primarily due to cash used in operations of $7.1 million offset by proceeds from sales under our ATM program of $1.4 million. Tempest ended the first quarter of 2022 with $45.8 million in cash and cash equivalents, compared to $51.8 million at December 31, 2021. On April 27, we announced a $15 million private investment in public equity (PIPE) financing to EcoR1 Capital, LLC and Versant Venture Capital. TREX1 Inhibitor (preclinical tumor-selective STING pathway activator): planned selection of development candidate in the second half of 2022.
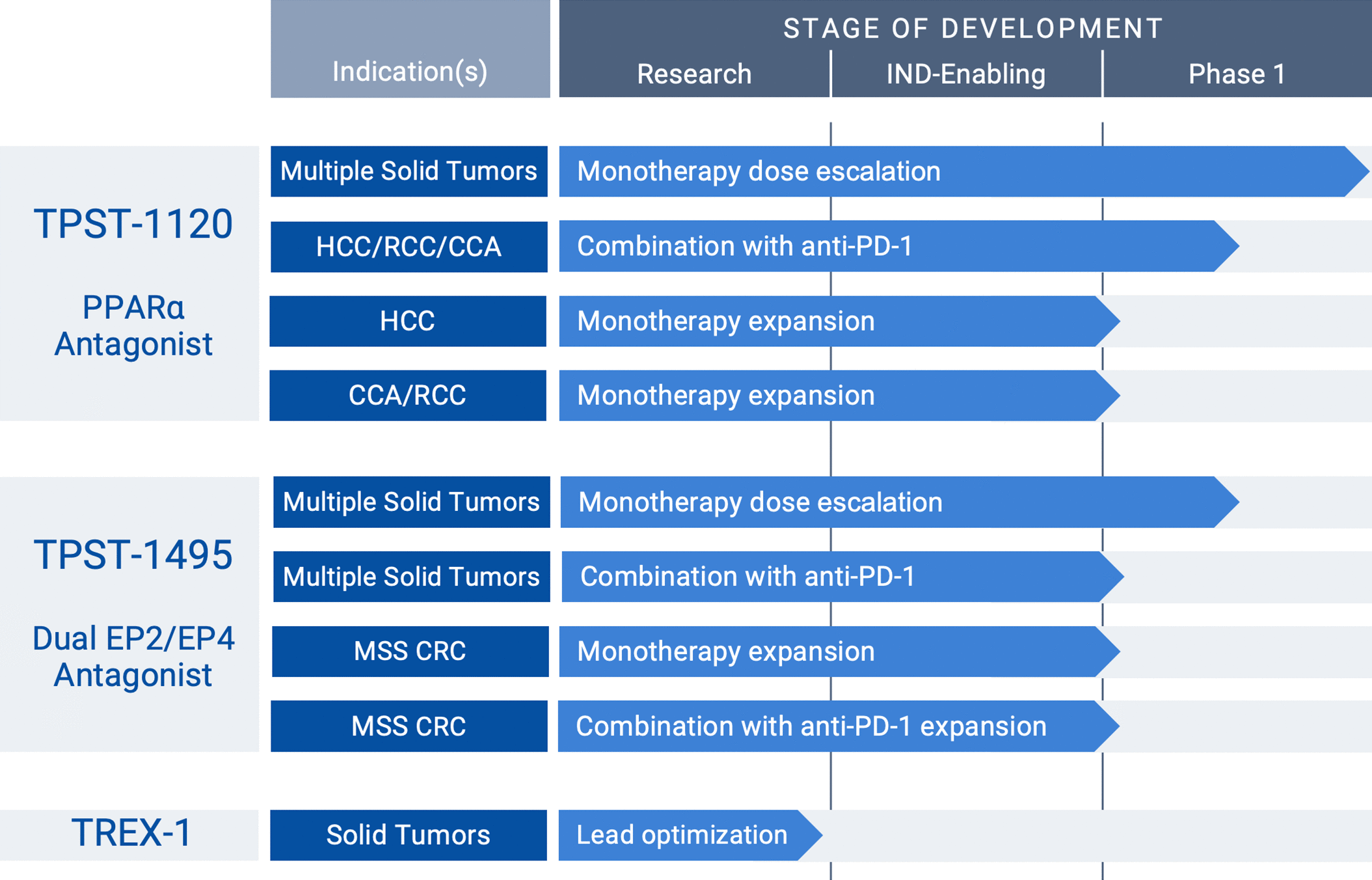
TPST-1495 (clinical dual EP2/4 prostaglandin receptor antagonist): (i) selection of monotherapy RP2D expected in the first half of 2022 (ii) presentation of “trial in progress” poster at ASCO 2022 Annual Meeting and (iii) data from Phase 1 monotherapy and combination dose and schedule optimization arms expected by year end or early 2023. TPST-1120 (clinical PPARα antagonist): (i) first presentation of clinical data from a Tempest program at the oral presentation of the Phase 1 monotherapy and combination data at the ASCO 2022 Annual Meeting and (ii) objective response data from the first 40 HCC patients in the first-line randomized study expected by year end or early 2023.

TREX1 Inhibitor (preclinical tumor-selective STING pathway activator): presented the first data with proprietary targeted molecules demonstrating therapeutic benefit in tumor-bearing mice at the AACR 2022 Annual Meeting. TPST-1495 (clinical dual EP2/4 prostaglandin receptor antagonist): (i) continued enrollment in a Phase 1 study evaluating both monotherapy and combination (with anti-PD-1 checkpoint inhibitor, pembrolizumab) dose and schedule optimization arms, towards establishing an RP2D (ii) presented preclinical data further differentiating TPST-1495 from other approaches targeting the prostaglandin E2 (PGE2) pathway at the American Association for Cancer Research (AACR) 2022 Annual Meeting and (ii) announced that ASCO accepted for a poster presentation a “trials in progress” for the ongoing TPST-1495 Phase 1 clinical monotherapy and combination therapy clinical trial. TPST-1120 (clinical PPARα antagonist): (i) completed enrollment in the Phase 1 monotherapy and combination dose escalation arms (ii) announced that the American Society of Clinical Oncology (ASCO) accepted for an oral presentation at its 2022 Annual Meeting an abstract containing the TPST-1120 Phase 1 monotherapy and combination data and (iii) continued enrollment in first-line, randomized global Phase 1b/2 study in patients with hepatocellular carcinoma (HCC), under a collaboration with F. “We look forward to the upcoming oral presentation, which will highlight the clinical profile and responses observed in the monotherapy and combination Phase 1 study of TPST-1120, our novel PPARa antagonist.” Brady, chief executive officer of Tempest. “The team continued to execute well throughout the first quarter of this year, enabling us to present the first clinical data from a Tempest program at the ASCO Annual Meeting in June,” said Stephen R. (Nasdaq: TPST), a clinical-stage oncology company developing first-in-class 1 therapeutics that combine both targeted and immune-mediated mechanisms, today reported financial results for the quarter ended Maand provided a corporate update. SOUTH SAN FRANCISCO, Calif., (GLOBE NEWSWIRE) - Tempest Therapeutics, Inc. Presented positive preclinical data with proprietary small molecule TREX1 inhibitors at AACR Presented positive preclinical data for TPST-1495 at AACR Announced oral presentation of TPST-1120 Phase 1 monotherapy and combination therapy data at ASCO
